The number of American kids diagnosed with ADHD has grown 30 percent in less than ten years. For parents looking to treat ADHD without medication, here’s some groundbreaking news. The U.S. Food and Drug Administration has just approved the first non-drug treatment for ADHD.
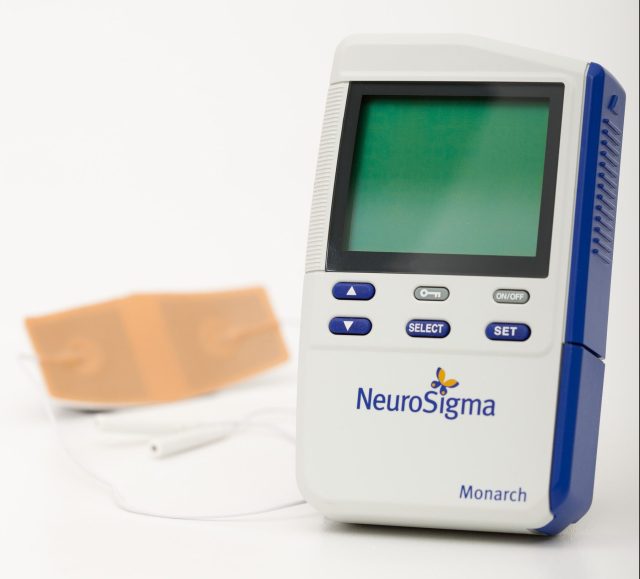
The new treatment is a medical device designed for use among kids seven to 12 who are not currently taking any ADHD prescription medication. About the size of a cell phone, the device dubbed the Monarch System “delivers low-level electrical stimulation via an external conductive patch on the patient’s forehead to the branches of the trigeminal nerve, which sends therapeutic signals to the parts of the brain thought to be involved in ADHD.”
photo: PRNewswire
Neuroimaging studies have shown that the device increases activity in the brain regions that are known to be important in regulating attention, emotion and behavior. It’s used primarily while sleeping and feels like a tingling sensation on the skin while in use. A prescription is required and clinical trails show it could take up to four weeks to see the effects. The common side effects include drowsiness, an increase in appetite, trouble sleeping, teeth clenching, headache and fatigue.
“This new device offers a safe, non-drug option for treatment of ADHD in pediatric patients through the use of mild nerve stimulation, a first of its kind,” said Carlos Peña, Ph.D., director of the Division of Neurological and Physical Medicine Devices in the FDA’s Center for Devices and Radiological Health. “Today’s action reflects our deep commitment to working with device manufacturers to advance the development of pediatric medical devices so that children have access to innovative, safe and effective medical devices that meet their unique needs.”
—Shahrzad Warkentin
RELATED STORIES
New Research Says More Screen Time Can Raise the Risk of an ADHD Diagnosis
The Number of Kids with ADHD in America Is Over 10 Percent—& It’s on the Rise
Study Examines How Modern Screens Like Smartphones & Tablets Impact Kids